-
Новости
- ИССЛЕДОВАТЬ
-
Страницы
-
Группы
-
Мероприятия
-
Статьи пользователей
-
Marketplace
-
Форумы
-
Игры
Clinical Trial Supplies Market Size, Share & Trends Analysis Report, 2030
Clinical Trial Supplies Market Growth & Trends
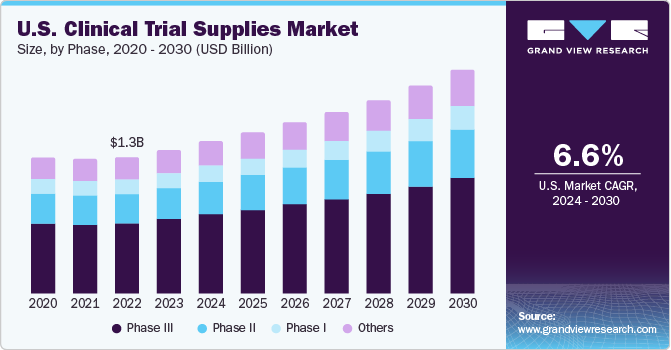
The global clinical trial supplies market size was estimated at USD 3.97 billion in 2030 and is anticipated to grow at a compound annual growth rate (CAGR) of 6.5% from 2024 to 2030. Increasing volume of clinical trial studies coupled with the growing complexity in conduction of these trials are some of the major factors driving the market growth.
Globally, an increase in the prevalence of chronic diseases and the rapidly aging population are expected to drive the growth of R&D of biologics, which is expected to further propel the demand for efficient clinical supplies and contribute to the growth of the clinical trial supplies industry. Furthermore, an increase in the demand for orphan drugs and high investment in the R&D of rare diseases are also expected to contribute toward the development of biologic drugs. Thus, owing to these factors, this segment is likely to witness significant growth during the forecast period.
For instance, in 2022, Novartis invested around USD 10 billion in research and development. It also secured 23 approvals in the European Union, Japan, China, and the U.S. for new drugs and rare diseases. The company is also conducting 44 ongoing phase III programs in India with 17 clinical programs running in rare diseases such as atypical hemolytic uremic syndrome (aHUS), Immune thrombocytopenic purpura (ITP), spinal muscular atrophy (SMA), and Lupus Nephritis.
Direct-to-Patients (DTP) is an upcoming segment in the distribution of clinical trial supplies, which is expected to be the future model of distribution. DTP is one of the emerging models that involves delivering drugs to patients directly to create patient-centric trials. This would facilitate fewer visits to the site and reduce the burden on participants. The COVID-19 outbreak has led to the increased adoption of such a model, to continue clinical trial studies with minimum disruption. In addition, patient retention and a diverse pool of patients worldwide are some of the notable reasons that can be attributed to the high adoption of this model.
Request a free sample copy or view report summary: https://www.grandviewresearch.com/industry-analysis/clinical-trial-supplies-market
Clinical Trial Supplies Market Report Highlights
- Based on the clinical phase, the market is anticipated to be dominated by the Phase III trial segment with a 52.7% revenue share in 2022. The presence of a large number of molecules currently under Phase III makes it the primary factor responsible for this deduction
- Among services, the storage, and distribution segment is anticipated to witness the fastest growth at a CAGR of 6.8% during the forecast period. The rise in global biologics pipeline and temperature-sensitive drugs is expected to increase the complexities related to the logistics of clinical trial supplies
- Biologicsare expected to witness the fastest growth at 6.7% CAGR during the forecast period owing to the increasing research in the field of genetics and biotechnology such as the development of nanoparticle-based drug delivery systems
- In terms of therapeutic use, oncology dominated the market with a revenue share of 38.8% in 2022. According to the United Press International, hospitals in the U.S. are disposing of billions of cancer drug vials due to improper dosage, thereby indicating the need for appropriate supply management
Clinical Trial Supplies Market Segmentation
Grand View Research has segmented the global clinical trial supplies market based on clinical phase, product/service, end-use, therapeutic use, and region:
Clinical Trial Supplies Clinical Phase Outlook (Revenue, USD Billion, 2018 - 2030)
- Phase I
- Phase II
- Phase III
- Others
Clinical Trial Supplies Product/Service Outlook (Revenue, USD Billion, 2018 - 2030)
- Manufacturing
- Storage & distribution
- Cold chain based
- Non-cold chain based
- Supply chain management
Clinical Trial Supplies End-Use Outlook (Revenue, USD Billion, 2018 - 2030)
- Pharmaceuticals
- Biologics
- Medical device
- Others
Clinical Trial Supplies Therapeutic Use Outlook (Revenue, USD Billion, 2018 - 2030)
- Oncology
- CNS
- Cardiovascular
- Infectious disease
- Metabolic disorders
- Others
Clinical Trial Supplies Regional Outlook (Revenue, USD Billion, 2018 - 2030)
- North America
- S.
- Canada
- Europe
- K.
- Germany
- France
- Italy
- Spain
- Denmark
- Sweden
- Norway
- Asia Pacific
- Japan
- China
- India
- Australia
- South Korea
- Thailand
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- South Africa
- Saudi Arabia
- UAE
- Kuwait
List of Key Players in the Clinical Trial Supplies Market
- Almac Group
- Biocair
- Catalent Inc.
- KLIFO
- Movianto
- PCI Pharma Services
- Sharp Services, LLC
- Thermo Fischer Scientific Inc.
- Marken
- PAREXEL International Corporation
Browse Full Report: https://www.grandviewresearch.com/industry-analysis/clinical-trial-supplies-market
- Whats New
- Shopping
- Wellness
- Sports
- Theater
- Religion
- Party
- Networking
- Music
- Literature
- Art
- Health
- Игры
- Food
- Drinks
- Fitness
- Gardening
- Dance
- Causes
- Film
- Crafts
- Other/General
- Cricket
- Grooming
- Technology

